Can You Flush With Water for Injection? Medical Procedures
Did you no that flushing medical devices with the correct fluid can significantly impact patient safety and treatment outcomes? In medical procedures, using Water for Injection (WFI) to flush intravenous lines or catheters is a common practice, but it raises critically important questions about when and how it should be used. This introduction explores whether you can safely flush with Water for Injection, clarifying its purpose, proper submission, and the critical role it plays in maintaining sterility and preventing complications. Understanding these nuances is essential for healthcare professionals and anyone involved in medical care to ensure safe practices and optimal patient outcomes. As you read on, you’ll discover practical insights into why this seemingly simple step demands careful attention and how it fits into broader clinical protocols. Whether you’re a medical practitioner or a curious learner, this discussion offers valuable knowledge to deepen your grasp of safe medical procedures.
Understanding Water for Injection: What It Is and Why It Matters
Water intended for injection plays a pivotal role in medical and pharmaceutical environments, distinguished by its unparalleled purity and stringent manufacturing standards. Unlike regular potable water,Water for Injection (WFI) is specially treated to be free from pyrogens,endotoxins,and microbial contamination,making it safe for use in sensitive clinical applications such as diluting medications or flushing devices that come into direct contact with sterile tissues or the bloodstream. This extraordinary level of purity is not incidental but the result of precise distillation or specialized purification processes mandated by pharmacopeial standards such as the United States Pharmacopeia (USP).
Understanding what sets WFI apart helps clarify why it matters so profoundly in medical procedures. For example, when flushing intravenous lines or catheters, using any contaminated or less pure fluid can introduce infections or inflammatory responses, jeopardizing patient safety.Thus, WFI serves as a critical safeguard against such risks due to its verified sterility and chemical purity.It is also important to note that WFI systems require rigorous maintenance, sanitation, and compliance with regulatory guidelines to ensure consistent quality over time-a fact underscored during pharmaceutical inspections focused on contamination control.
Key Characteristics of Water for Injection
- Purity: Free from endotoxins, bacteria, particulates, and chemical contaminants.
- Manufacturing Standards: Produced and stored under controlled conditions,often involving high-temperature storage or periodic sanitization to prevent microbial growth.
- Regulatory Compliance: Adheres to strict USP monographs with detailed specifications to ensure safety and efficacy.
Given these attributes, WFI is not just “clean water” but a pharmaceutical-grade utility that supports both drug formulation and medical device maintenance. Its use in flushing is often preferred or mandated precisely because it minimizes the risk of adverse reactions, supports infection control, and maintains the integrity of medical equipment. For clinicians and healthcare providers, appreciating these factors is essential to optimizing patient outcomes and complying with established health standards.By choosing Water for Injection for flushing procedures, healthcare facilities affirm thier commitment to the highest standards of care, ultimately ensuring that even seemingly simple tasks, like flushing a device, do not become avenues for contamination or harm. This makes understanding WFI’s unique qualities essential for anyone involved in medical device use or pharmaceutical compounding.
Medical Guidelines on flushing Procedures: Safe Practices Explained
Flushing medical devices such as intravenous (IV) lines, catheters, and PICC lines is a routine yet critical procedure that directly influences patient safety and treatment efficacy. Using the correct flushing fluid and adhering to established protocols can prevent serious complications like infections, occlusions, or device malfunctions. In this context, guidelines emphasize the importance of careful handling and readiness to maintain a sterile environment while ensuring the device’s patency.
When flushing with Water for Injection (WFI) or other solutions like normal saline, healthcare professionals must strictly follow aseptic techniques. This includes using sterile syringes, disinfecting connection sites with appropriate antiseptics such as alcohol swabs, and employing the recommended volume and flushing method tailored to the device type. such as, a common approach used by nurses involves the “push-pause” technique-gently injecting fluid in bursts rather than a continuous force-to create turbulence that dislodges debris or clots effectively without damaging the vessel or device lining. Routine flushing intervals also play a role; many protocols call for flushing before and after medication administration and at regular intervals (e.g., every 12 hours) when the device is not in use to minimize blockage risks.
- Ensure Compatibility: Verify that the flushing solution is appropriate for the device and compatible with any medications previously administered to avoid adverse reactions or precipitation.
- Volume and Pressure Control: Use the correct volume as prescribed (typically between 3-10 mL) and avoid excessive pressure which can rupture catheters or cause vein irritation.
- Maintain sterility: Always inspect syringes and fluid containers for contamination,and never reuse disposable components.
For home-based patients managing lines such as PICC, education on flushing procedures is essential. Providers frequently enough supply saline or WFI syringes with clear instructions on timing and technique, helping reduce infection risks outside the clinical environment. Real-world practices also highlight that some healthcare settings prefer normal saline over WFI for flushing due to cost and availability, but only when strict infection control guidelines and standards support this choice. Ultimately, the safest approach always aligns with institutional protocols and clinical judgment, prioritizing patient safety and device longevity.
Practical Example: Flushing a PICC Line at Home
A typical home care instruction for PICC line flushing may include:
| Step | Recommended Action |
|---|---|
| 1. Preparation | Wash hands thoroughly and wear gloves. |
| 2.Disinfect | Clean the PICC line access port with an alcohol swab for 15 seconds and let dry. |
| 3. Attach Syringe | Use a pre-filled sterile syringe with saline or WFI as directed. |
| 4. Flush Using Push-Pause Technique | Slowly inject small amounts of fluid in short bursts to clear the line. |
| 5. Clamp and Disconnect | Clamp the line if applicable and safely remove the syringe. |
| 6.Dispose | Discard syringes and gloves properly; perform hand hygiene. |
Adhering to these systematic steps minimizes infection risk and ensures the device functions properly between treatments. With increasing patient self-management, reinforcing such safe practices through clear guidance and regular healthcare follow-up is indispensable.
medical guidelines on flushing procedures underscore a multi-faceted approach combining solution selection, technique precision, infection control, and patient education. Whether in hospital settings or home care, these practices safeguard against complications and uphold the standard of care essential when flushing with Water for Injection or option fluids.[1]
[2]
[3]
Can You Use Water for Injection to Flush Medical Devices?
Water for Injection (WFI) holds a unique place in medical procedures due to its exceptional purity and sterility, making it a highly controlled resource primarily designed for pharmaceutical and clinical use. Unlike sterile water, which contains no solutes and can cause osmotic damage to blood cells if used improperly, Water for Injection is carefully produced to meet strict pharmacopoeial standards ensuring it is free from pyrogens, endotoxins, and contaminants. This level of purity qualifies it as safe for flushing certain medical devices, but its application must be precise and clinically justified.
In practice, WFI is commonly used to flush medical devices such as intravenous lines, central venous catheters, and PICC lines, particularly when other flushing solutions like saline are contraindicated or unavailable. However, it is essential to consider that WFI lacks electrolytes and buffers, so its use as a flushing fluid might be limited by the clinical situation.For example, flushing a device with WFI is appropriate when clearing medication residua or preparing the line for therapies requiring absolutely no ionic interference.Nevertheless, because WFI is hypotonic, repeated or high-volume flushing might lead to shifts in fluid balance or cellular irritation if it enters the bloodstream directly.
When and How to Use WFI for Flushing Medical Devices
- Ensure Device Compatibility: Some catheters and tubing materials react differently to solutions. Confirm that the device manufacturer approves WFI as a flushing agent to prevent material degradation.
- Use Aseptic Technique: WFI syringes and containers should remain sealed until use, handled with clean gloves to avoid contamination, and discarded after single use.
- Appropriate Volume and Frequency: Follow institutional protocols that specify volume (typically 2-10 mL) and frequency, such as flushing before and after medication administration or every 12 hours when inactive.
- Consider Patient Factors: For patients with electrolyte or fluid imbalances, consult healthcare providers before using WFI for flushing to avoid unintended clinical effects.
While saline is favored in many clinical settings due to its isotonic properties and electrolyte content, WFI remains indispensable in scenarios demanding the utmost purity and absence of additives. As a notable example, it is indeed the flushing fluid of choice when administering or flushing lines with medications incompatible with saline or when preparing intravenous mixtures during sterile compounding. Additionally, WFI is ideal for flushing implantable ports or devices sensitive to saline residues.
Water for Injection can be a safe and effective flushing option for medical devices when used correctly, respecting its properties and limitations. The best outcomes emerge from combining strict aseptic technique, adherence to clinical protocols, and individualized patient care. Healthcare providers should remain informed about the distinctions between flushing fluids to optimize device maintenance and patient safety.
Risks and Consequences of Improper Flushing Solutions
Even seemingly minor missteps in selecting or administering flushing solutions can lead to significant clinical complications. using inappropriate fluids-such as sterile water instead of Water for injection (WFI) or saline-can cause adverse reactions ranging from cellular damage to infection risks. The osmolarity of the flushing solution plays a pivotal role; as a notable example, sterile water has an osmolarity of zero and can induce hemolysis if it enters the bloodstream directly, as red blood cells swell and rupture due to osmotic imbalance. This potent danger underscores why sterile water should never be used as a flush solution in intravenous lines or catheters[[[[[2]](https://allnurses.com/flushing-sterile-water-t562304/).
Key Risks Associated with Improper Flushing Solutions
- Cellular and Tissue Damage: Hypotonic solutions like sterile water may cause hemolysis or damage to the vascular endothelium, leading to inflammation or thrombosis.
- Device Material Degradation: Some catheters and implantable ports are sensitive to certain flushing agents. Using non-approved solutions can weaken device integrity, possibly causing leaks or breakage.
- Risk of Contamination and Infection: Non-sterile or improperly handled flushing fluids can introduce pathogens, increasing the risk of bloodstream infections-especially in immunocompromised patients.
- Electrolyte Imbalance and Clinical Instability: Flushing solutions lacking electrolytes or buffers may disrupt a patient’s fluid and electrolyte balance if used repeatedly or in large volumes, potentially exacerbating pre-existing conditions.
Choosing the right flush solution is not only about the chemical composition but also about respecting procedural protocols. Such as, Water for Injection is highly purified and free from pyrogens and endotoxins, making it suitable for certain flush applications where additive-free flushing is necessary. However, even WFI must be used cautiously-excessive volume or frequency can lead to adverse systemic effects due to its hypotonic nature. Healthcare providers should always verify device compatibility and adhere strictly to aseptic technique to minimize risk.
| Flushing Fluid | Typical Use | Risk if Used Improperly |
|---|---|---|
| Water for injection (WFI) | Flushing medication residue, preparing lines for sensitive drugs | Fluid shifts; irritation if overused or improperly administered |
| Sterile Water | Not recommended for flushing blood-contact lines | Hemolysis and vascular damage due to osmotic imbalance |
| Normal Saline (0.9% NaCl) | Standard flushing fluid; isotonic and electrolytes included | Limited if incompatible with medication; possible fluid overload in susceptible patients |
In real-world clinical settings, misunderstandings about flushing fluids are common, with some practitioners mistakenly opting for sterile water or tap water in emergencies. Such practices can lead to complications ranging from immediate hemolysis to long-term device failure or bloodstream infections. Thorough training and accessible, clear protocols are essential to ensure the right product is used every time.
Ultimately, awareness about the physiochemical properties of flushing solutions and strict compliance with medical guidelines protect both patient safety and device functionality. When in doubt, consulting current standards and device manufacturer recommendations is the best course of action to avoid preventable adverse outcomes.
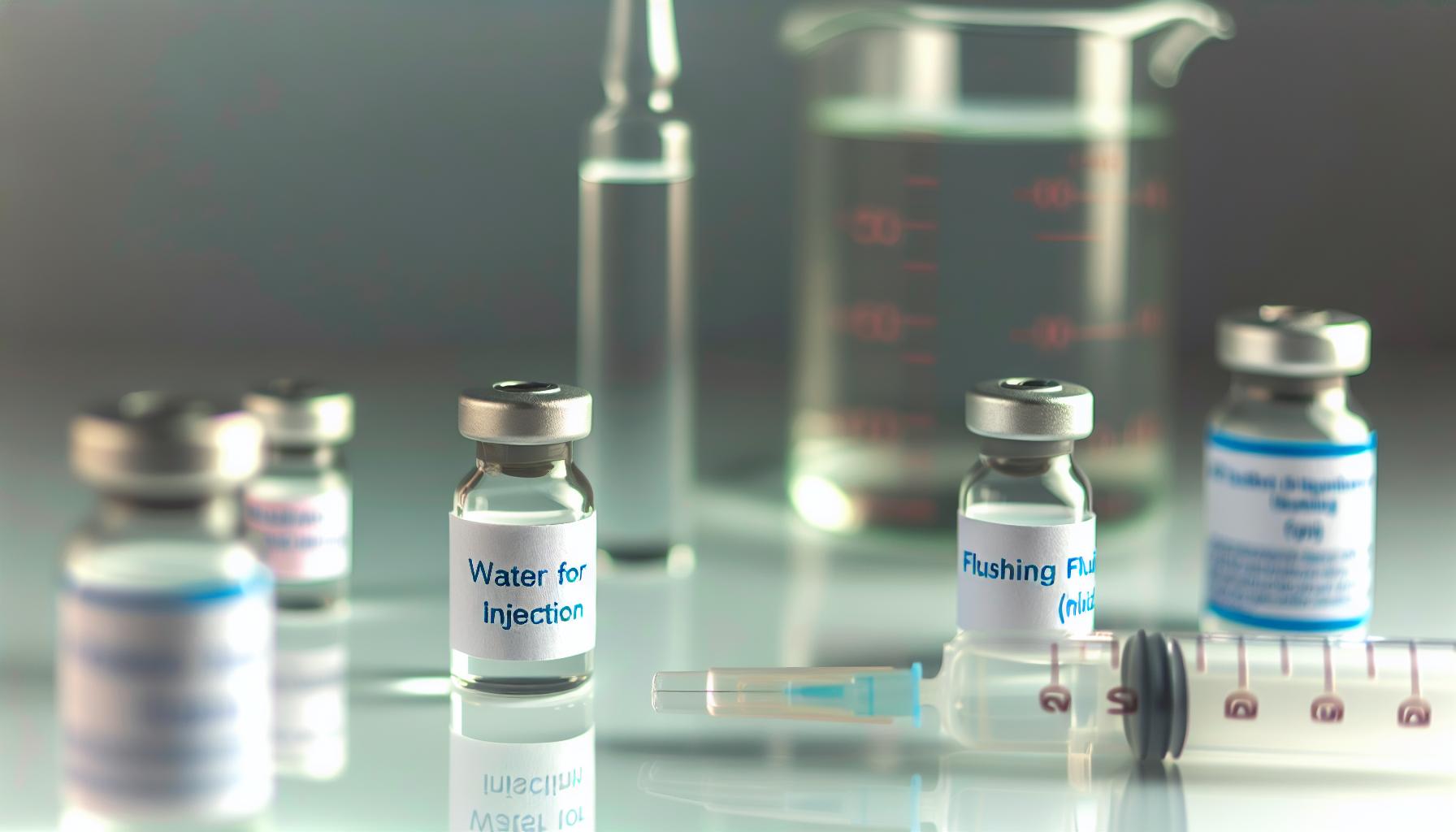
Comparing Water for Injection and Other Flushing Fluids
Flushing solutions play a critical role in maintaining IV line patency, preventing contamination, and ensuring medication efficacy. Among these, water for Injection (WFI) stands out due to its exceptional purity and sterility, making it a preferred choice in specific circumstances. However, it’s important to weigh its properties against other commonly used flush fluids like normal saline and sterile water to understand where each fits best in clinical practice.
WFI is ultra-purified, free from endotoxins and pyrogens, and generated under stringent regulatory standards.This makes it ideal for flushing medication residues or preparing lines for sensitive drugs that could react adversely with electrolytes or other additives found in saline. Yet, WFI’s lack of electrolytes also renders it hypotonic, meaning it has the potential to cause fluid shifts if used excessively or improperly within blood-contact lines. In contrast, normal saline is isotonic, closely matching the osmolarity of human plasma, and thus is generally safer for routine flushing to avoid cellular damage and maintain electrolyte balance.
Sterile water, while also highly purified, differs significantly because it lacks both electrolytes and the rigorous endotoxin control of WFI. This makes it inappropriate for flushing intravascular devices due to its risk of causing hemolysis and vascular endothelial irritation from osmotic imbalance. Despite some misconceptions, using sterile water as a flush can lead to harm rather than help, highlighting why the choice of solution should never be based on assumptions but on clear evidence and device compatibility recommendations.
Practical Differences and Considerations
- purity and Safety: WFI undergoes more stringent purification, ensuring it’s free from pyrogens and endotoxins-critical in sensitive clinical situations.
- Osmolarity and Cellular Impact: Normal saline, being isotonic, minimizes the risk of cellular damage, while WFI (hypotonic) must be used cautiously.
- Device Compatibility: Some catheter materials may degrade with certain fluids; confirming compatibility with WFI or saline is essential.
- Clinical Use Cases: WFI works well for flushing medication lines where additives might cause precipitation,whereas saline is better suited for general maintenance flushing.
| Flushing Fluid | Key Characteristics | Ideal Use | Main Caution |
|---|---|---|---|
| Water for Injection (WFI) | Extremely pure, pyrogen-free, endotoxin-free, hypotonic | Flushing medication residue, sensitive drug prep, wound irrigation | Risk of fluid shifts/irritation if overused intravenously |
| Normal Saline (0.9% NaCl) | Isotonic, contains electrolytes, widely available | Routine IV flushing, maintaining catheter patency | Potential incompatibility with certain meds, fluid overload risk |
| Sterile Water | Pure but lacks endotoxin control, hypotonic | Not recommended for IV flushing, sometimes used externally | Hemolysis risk, vascular irritation |
Understanding these differences empowers healthcare professionals to select the safest and moast effective flush solution tailored to each clinical scenario. For example, using WFI to flush an IV line before administering a sensitive chemotherapy agent can prevent unwanted reactions caused by residual electrolytes in saline. Conversely,for routine maintenance of peripheral lines,saline’s physiological compatibility frequently enough makes it the better choice.Knowing when and how to use these solutions helps prevent serious complications such as hemolysis or infection and promotes optimal patient outcomes.
Ultimately, comprehensive training, adherence to updated protocols, and attention to device manufacturer instructions are key to ensuring flushing practices are both effective and safe. In practice, this means reserving WFI for situations demanding the highest purity and avoiding sterile water for intravascular flushes to prevent avoidable harm.
Step-by-Step Guide: Proper Techniques for Flushing with Water for Injection
Flushing with Water for Injection (WFI) requires precision and awareness due to its unique properties compared to other flushing solutions. As WFI is extremely pure and free from endotoxins and pyrogens, it’s ideally suited for clearing medication residues from lines used for sensitive drugs. However, its hypotonic nature means that an improper flushing technique can lead to unwanted osmotic effects, so careful handling is essential to maintain patient safety and device integrity.
Before starting, ensure all equipment is sterile and compatible with WFI. Use aseptic technique to prevent contamination. prepare the flush syringe by drawing the exact prescribed volume of WFI, avoiding air bubbles that can cause embolism or inaccurate dosing. When connecting to the catheter or IV line, verify that the material is compatible with WFI to prevent degradation over time. Not all catheters are designed to tolerate the unique characteristics of WFI, so always follow manufacturer guidelines.
Performing the Flush
- Step 1: Patency Check
Gently aspirate to confirm catheter patency and ensure the line is free of blood clots or obstructions. - Step 2: slow and Steady Injection
Administer the WFI flush slowly, typically over 10 to 30 seconds, depending on the catheter size and patient condition.Rapid injection might induce fluid shifts or irritation due to hypotonicity. - Step 3: monitor Patient Response
Observe the patient closely during and after flushing for any signs of discomfort, swelling, or infiltration. Because WFI lacks electrolytes, excessive use or inappropriate dosing could lead to local tissue irritation or cellular fluid imbalances. - Step 4: Document Thoroughly
Record the flushing procedure details, including volume used, time, patient tolerance, and any adverse reactions.
Additional Tips for Enhanced safety and Efficacy
- Limit the use of WFI flushes primarily to scenarios where saline or other isotonic solutions might interact negatively with medications or device materials.
- Never substitute sterile water for WFI when flushing intravascular lines; sterile water lacks endotoxin control and can cause hemolysis and vessel irritation.
- When flushing after medications prone to precipitation, a WFI flush can help clear residual drug particles to reduce catheter occlusion risks.
- Regularly train clinical staff on the differences between flushing solutions to reinforce safe and evidence-based practices.
| Flush Step | Key Points |
|---|---|
| Preparation | Use sterile technique; confirm WFI and device compatibility; avoid air bubbles. |
| Administration | Inject slowly; monitor patient vitals; avoid rapid bolus to prevent irritation. |
| Post-flush | Observe for adverse effects; document procedure and patient response. |
Following these best practices ensures that flushing with Water for Injection effectively maintains line patency and medication efficacy while safeguarding patient health. Understanding both the power and limitations of WFI as a flush allows clinicians to use it confidently and optimally within the broader range of flushing protocols.
Clinical Scenarios Where Flushing with Water for Injection Is Recommended
water for Injection (WFI) holds a unique position in clinical practice thanks to its exceptional purity and stringent manufacturing standards. This makes it an indispensable flushing agent in situations where even minimal contaminants or electrolytes could compromise the safety and effectiveness of treatment. As an example, WFI is especially recommended when dealing with lines previously used for sensitive or reactive drugs that might precipitate or degrade in the presence of saline or other isotonic solutions. Flushing with WFI can help ensure that no residual drug remains,reducing the risk of catheter occlusion or adverse reactions.In addition,WFI is often the flush of choice in neonatal and pediatric care,where the delicate physiology of patients demands the use of ultra-pure fluids with no added electrolytes that could disturb delicate fluid balances. Flushing with WFI in these scenarios supports maintaining line patency without introducing ionic imbalances. Similarly, in immunocompromised patients such as those undergoing chemotherapy or bone marrow transplantation, the sterility and absence of pyrogens in WFI make it a safer flushing option to minimize infection risks and irritation.
Common Clinical Indications for Using Water for Injection Flushes
- Post-Administration of Precipitation-Prone drugs: Some antibiotics, chemotherapeutic agents, and contrast media can crystallize or interact unpredictably with saline.A WFI flush effectively clears these residuals.
- Prior to Sensitive Biologic or Enzyme Therapy: To avoid ionic interference with delicate biologics, flushing with WFI helps maintain the integrity of catheters and delivery systems.
- When Electrolyte-Free Flushing Is Required: Certain diagnostic and therapeutic procedures call for electrolyte-free solutions to prevent interactions or vessel irritation.
- During Device Priming and Maintenance: WFI is used to prime intravenous lines and infusion pumps to ensure no contaminants disrupt flow or patient outcomes.
While WFI’s hypotonic nature requires cautious administration, its use in these carefully selected clinical contexts enhances patient safety and device performance. Clinicians must, however, always verify catheter compatibility and patient condition before opting for a WFI flush, to prevent complications such as hemolysis or local tissue irritation. Proper training and awareness remain critical to leveraging the advantages of WFI flushes effectively within clinical protocols.
Common Myths and Misconceptions About Flushing with Water for Injection
Water for Injection (WFI) often sparks confusion due to its deceptively simple nature-it’s just pure water, after all-but in clinical settings, its role is anything but simple. One common misconception is that WFI is interchangeable with saline or sterile water for irrigation when flushing medical devices. While all are sterile, WFI’s unique preparation standards ensure it’s free from endotoxins and particulates, making it significantly safer in scenarios requiring utmost purity. Using other fluids without considering these differences can lead to complications such as irritation, catheter occlusion, or inaccurate dosing.
Another prevalent myth is that flushing with WFI carries no risks because it’s just water. In reality, WFI is hypotonic-meaning it lacks electrolytes-so improper or excessive use, especially in vascular lines, may cause hemolysis or local endothelial irritation. Its hypotonicity demands cautious administration and strict adherence to clinical guidelines to avoid adverse outcomes. Such as,neonates or patients with fragile veins require meticulous monitoring when flushing with WFI to prevent imbalances or damage.
Many practitioners also assume that WFI can always replace saline or heparin flushes. however, these fluids serve distinct purposes: saline flushes primarily clear the line and confirm patency, while heparin flushes prevent clot formation. WFI,due to its electrolyte-free composition,is mainly indicated where ionic interference must be avoided or in device preparation and cleaning. Mistaking one flush for another without appreciating these roles can compromise treatment effectiveness and patient safety.
Practical Tips to Navigate Flushing Choices
- always consult institutional protocols: Many facilities have strict guidelines defining when WFI is appropriate versus saline or heparin flushes.
- Verify patient condition and catheter type: Tailor flush choices to clinical context, such as patient age, immune status, and device compatibility.
- Train staff on subtle differences: Educate healthcare providers on hypotonic effects of WFI and proper flushing techniques to avoid misuse.
- Use WFI primarily when indicated: Reserve for cases involving sensitive drugs, neonatal care, or device priming where electrolyte absence is critical.
Understanding these nuances not only prevents the common pitfalls surrounding WFI use but also enhances overall flushing safety, ensuring patients benefit from both the purity of WFI and the protective functions of other flushing agents when needed.
Regulatory Standards and Compliance for Using Water for Injection
water for Injection (WFI) is governed by some of the most stringent regulatory standards in pharmaceutical and healthcare settings due to its critical role in patient safety. Unlike potable or sterile water, WFI must meet rigorous purity criteria to ensure it is indeed free from endotoxins, particulates, and microbial contamination, as it is frequently used in situations requiring the highest level of sterility-such as drug reconstitution, device flushing, and intravenous administration. Regulatory bodies like the U.S. Pharmacopeia (USP) and the FDA outline precise manufacturing and testing protocols for WFI to safeguard against contamination risks that could jeopardize patient outcomes.
The USP sets forth detailed monographs specifying WFI quality parameters-including limits for total organic carbon,conductivity,endotoxin content,and microbial bioburden-that manufacturers and healthcare providers must adhere to strictly. Additionally, the production process often involves distillation, reverse osmosis, or other validated purification technologies coupled with routine system sanitization to maintain water integrity. Regulatory guidelines emphasize that water systems used to supply WFI must include corrective facilities enabling steam sterilization, chlorination, or filtration, thus guaranteeing consistent compliance throughout the supply chain[[[[[1]](https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-technical-guides/water-pharmaceutical-use).
Ensuring Compliance in Clinical Practice
Maintaining regulatory compliance extends beyond the manufacturing environment and into clinical usage. hospitals and healthcare providers must implement strict protocols for handling and storing WFI to prevent contamination before use. For example, WFI should be stored in closed, sanitized, and temperature-controlled containers to prevent endotoxin proliferation. When flushing medical devices, healthcare staff must ensure that only WFI batches meeting pharmacopeial standards are used, following institutional policies that align with regulatory mandates.
Many institutions mandate documentation and traceability of WFI batches used in patient care to facilitate quality assurance and enable swift action if deviations or adverse events arise.compliance audits frequently assess whether WFI handling-including transfer to syringes or flushing systems-follows aseptic techniques and validation procedures. Since improper use of non-compliant water can lead to severe risks such as infection or device malfunction, ongoing staff training on regulatory expectations is essential.
Guidelines to Navigate Compliance Challenges
- Validate water purification systems: Conduct regular testing and maintenance per USP and FDA guidelines to ensure WFI purity.
- Adhere to storage protocols: Use endotoxin-free containers and avoid prolonged storage or exposure to conditions favoring microbial growth.
- Follow aseptic technique during use: Ensure that handling of WFI for flushing or drug preparation minimizes contamination risk.
- Maintain comprehensive documentation: record batch numbers, expiration dates, and usage details for traceability and quality control.
Hospitals should also stay abreast of updates to pharmacopeial standards and regulatory guidance, as advances in water system technologies and microbial detection methods continue to evolve. This proactive approach helps minimize risks linked to endotoxins or impurities that could compromise patient safety during medical device flushing or other clinical applications.
By integrating robust regulatory compliance into both water generation and clinical use practices, healthcare providers can confidently utilize Water for Injection with assurance of its quality, efficacy, and safety-ultimately enhancing patient care outcomes and fulfilling critical quality standards in medical procedures[[[[[1]](https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-technical-guides/water-pharmaceutical-use).
Expert Tips to Ensure Safety and Efficacy When Flushing
Water for Injection (WFI) is a vital asset in ensuring the sterility and safety of flushing procedures, but its benefits can only be fully realized through careful and educated handling. Healthcare providers and caregivers must recognize that even the purest WFI can become compromised if not used correctly. A key insight is that maintaining asepsis during flushing not only preserves the integrity of medical devices but also significantly reduces the risk of introducing infections.
- Confirm Source and Integrity: Always verify that the WFI used is from a validated, sterile source and stored according to recommended conditions. Containers should be sealed and free from contamination risks, with expiry dates closely monitored. Never use WFI that has been improperly stored or has passed its quality assurance checks.
- Use Proper Technique: Flushing protocols must strictly follow aseptic procedures. This includes using sterile gloves, employing single-use syringes or equipment for each flush, and minimizing the time the WFI container remains open. Proper flushing volumes and flow rates, as prescribed by clinical guidelines, are crucial to preventing device occlusion or damage while ensuring adequate clearance.
- Document Thoroughly: Keeping detailed records of WFI batch numbers, expiration, and usage times is essential for traceability.this practice aids in identifying potential sources of contamination or adverse events and supports compliance with regulatory audits.
- Regular Staff Training: Even experienced clinicians can benefit from ongoing education about the unique properties of WFI and evolving standards for its use. Training ensures awareness about potential pitfalls such as endotoxin contamination and emphasizes the importance of handling techniques that maintain solution purity.
Practical Example: Preventing Contamination During Flushing
Consider a scenario in a hospital infusion unit where a PICC line requires regular flushing between medication administrations.If WFI is drawn into a syringe but then left uncapped on a tray for an extended period, airborne contaminants and endotoxins could infiltrate the solution, negating the inherent sterility of WFI.Using a new syringe and freshly drawn WFI for each flush with proper aseptic handling preserves patient safety and device functionality.
Healthcare settings can also implement routine audits of flushing protocols and WFI handling practices to detect and address any deviations promptly. This proactive approach reduces risks and upholds best practices.
By integrating these expert safeguards and considering the nuances of WFI handling, clinicians ensure that flushing delivers maximum therapeutic benefit without compromising patient well-being. The combination of high-quality water,impeccable technique,and stringent documentation forms the backbone of safe and effective flushing in medical procedures.
Emerging Research and Innovations in flushing Practices
Innovations in flushing practices continue to evolve as healthcare increasingly embraces advanced sterilization and safety protocols. One of the most fascinating developments is the integration of automated flushing systems that utilize precisely measured Water for Injection (WFI) doses, which minimize human error and reduce contamination risks.These systems ensure that flushing occurs consistently at optimal flow rates and volumes tailored to specific medical devices, enhancing device longevity and patient safety.
Moreover, research explores combining WFI with antimicrobial agents specially formulated to maintain sterility without causing device damage. While WFI alone is the gold standard for purity, ongoing studies are investigating additives that could provide an extra defensive layer against biofilm formation-an frequently enough arduous-to-treat issue in indwelling catheters and central lines. Such advancements could transform flushing from a purely mechanical process into a preventative strategy against device-associated infections.
Emerging Practical Approaches
- Closed-System Flushing: Newly designed closed systems protect the flushing solution from environmental exposure, drastically lowering endotoxin contamination risks. These systems simplify aseptic technique, making them ideal for both hospital and home care settings.
- Smart monitoring Technologies: Devices with integrated sensors can track flushing frequency, volume, and timing, alerting healthcare providers when protocols deviate.This real-time monitoring enhances compliance with flushing recommendations, ensuring WFI is used effectively and safely.
- Personalized Flushing Protocols: With increasing emphasis on patient-specific care, protocols are being tailored based on individual patient risk factors such as immune status, device type, and therapy duration. This personalized approach optimizes the use of WFI, avoiding unnecessary flushing or insufficient clearance.
These innovations illustrate a trend toward combining the exceptional purity of WFI with technology-driven solutions and evidence-based protocols.As an example, a hospital recently adopted an automated flushing device for PICC lines that utilizes WFI stored in sterile, single-use cartridges. Early results showed a marked reduction in catheter occlusions and a drop in infection rates. Such outcomes highlight how adopting cutting-edge methods not only improves clinical results but also streamlines workflows and enhances patient comfort.
In the near future, we can expect even greater integration of biotechnology, such as coatings on device lumens activated by flush solutions or WFI infused with nanoscale agents designed to disrupt microbial adhesion. Staying abreast of these developments empowers clinicians and caregivers alike to maintain the highest standards in medical device care while leveraging Water for injection’s unrivaled purity as the cornerstone of effective flushing.
Faq
Q: How does flushing with Water for Injection affect catheter patency in medical procedures?
A: Flushing with Water for Injection (WFI) helps maintain catheter patency by preventing blockages and minimizing residue buildup.Its sterile, pyrogen-free quality ensures safety during flushing. For optimal results, follow proper flushing techniques as outlined in the Step-by-Step Guide: Proper Techniques for Flushing with Water for Injection section to enhance device longevity and patient safety.
Q: When should Water for Injection be preferred over saline for flushing in clinical settings?
A: Water for Injection is preferred over saline in situations requiring non-ionic, pyrogen-free flushing, such as before drug infusion or when saline may cause precipitation. Use WFI when avoiding electrolyte interference is crucial-refer to Clinical Scenarios Where Flushing with Water for Injection Is Recommended for detailed guidance on specific use cases.
Q: What precautions should be taken to avoid contamination when flushing with Water for Injection?
A: To avoid contamination,always use aseptic technique during flushing with Water for Injection-use sterile equipment and avoid contact with non-sterile surfaces. Ensure strict compliance with manufacturer and regulatory standards detailed in Regulatory Standards and Compliance for Using Water for Injection to maintain safety and efficacy.
Q: Can flushing with Water for Injection cause damage to vascular tissues or implants?
A: Flushing with water for Injection is generally safe and non-irritating when used correctly. However, excessive or forceful flushing can potentially damage delicate vascular tissues or sensitive implants. Follow recommended techniques found in Expert Tips to Ensure Safety and Efficacy When Flushing to minimize risks.
Q: How do healthcare providers verify the sterility and purity of Water for Injection used in flushing?
A: Healthcare providers verify WFI sterility by sourcing it from certified pharmaceutical-grade suppliers and checking batch certifications. Routine quality control and adherence to guidelines in the Regulatory Standards and Compliance for Using Water for Injection section are essential to ensure purity before clinical use.
Q: What are the differences in patient outcomes when flushing with Water for Injection versus other fluids?
A: Patients flushed with Water for Injection often experience reduced risks of infection and irritation compared to other fluids, thanks to WFI’s purity and lack of additives. For comprehensive comparisons, consult Comparing Water for Injection and Other Flushing Fluids, which highlights outcomes and safety profiles.
Q: Why is Water for Injection not always suitable for flushing all medical devices?
A: Water for Injection may not be suitable when osmotic balance or ion content is critical, such as in devices sensitive to hypotonic fluids. Always evaluate device compatibility outlined in Medical Guidelines on Flushing Procedures: Safe Practices Explained before deciding to use WFI for flushing.
Q: How should leftover Water for injection be stored or disposed of after flushing procedures?
A: Leftover Water for Injection should be stored in sterile, sealed containers and used promptly to prevent contamination. If disposal is necessary, follow institutional protocols for pharmaceutical waste as detailed in Regulatory Standards and Compliance for Using Water for Injection to ensure environmental safety.
For deeper insights and detailed techniques, explore the full guidance in our article sections on flushing procedures and safety.
To Wrap It Up
Understanding the appropriate use of water for injection in medical procedures is crucial for ensuring patient safety and effective treatment outcomes. by following best practices for flushing, you minimize risks of contamination and optimize procedural success. If you’re looking to deepen your knowledge, explore our detailed guides on aseptic techniques and intravenous therapy protocols to enhance your clinical confidence.
Ready to apply these insights? Subscribe to our newsletter for the latest updates on medical procedures, or schedule a consultation with our experts to discuss tailored approaches for your practice. Don’t hesitate to ask questions or share your experiences in the comments-your engagement helps us build a trusted community focused on safe and effective healthcare practices. For more on related topics like medication administration and infection control, continue browsing our resource library and stay informed on the best standards in care.